
Solved RT B 2. The compressiblity factor for a gas is
4.6 (578) In stock

4.6 (578) In stock
Answer to Solved RT B 2. The compressiblity factor for a gas is

Solved] The Redlich-Kwong equation of state is gi

The equation of state of a real gas is p(v-b) =RT Can the gas be liquefied? Explain? - EduRev Class 11 Question

Thermodynamics Review Problems, PDF, Entropy

The compressibility factor of a van der Waals gas the critical point is equal to

COMPRESSIBILITY FACTOR

Non-Ideal Gas Behavior Chemistry: Atoms First
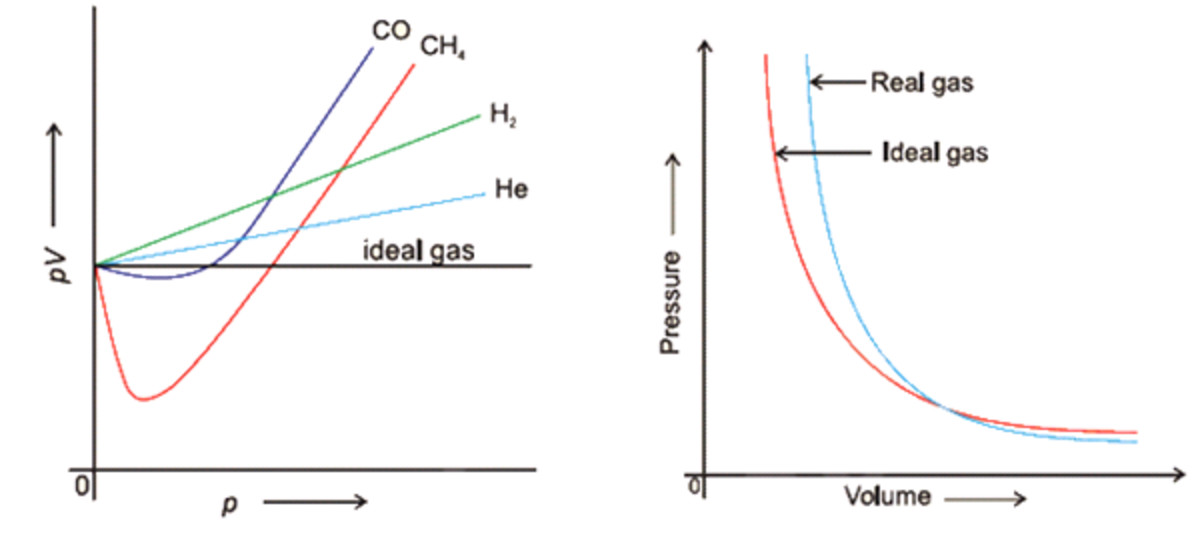
Real Gases - Chemistry, Class 11, States of Matter

Real gasses For an ideal gas, the compressibility factor Z = PV/nRT is equal to unity for all conditions. For a real gas, Z can be expressed as a function. - ppt

Real gases

solution manual for applied petroleum reservoir engineering by

OneClass: For a real gas, the compressibility factor, Z, is defined as Z (T, P) = PV/nRT For an ideal
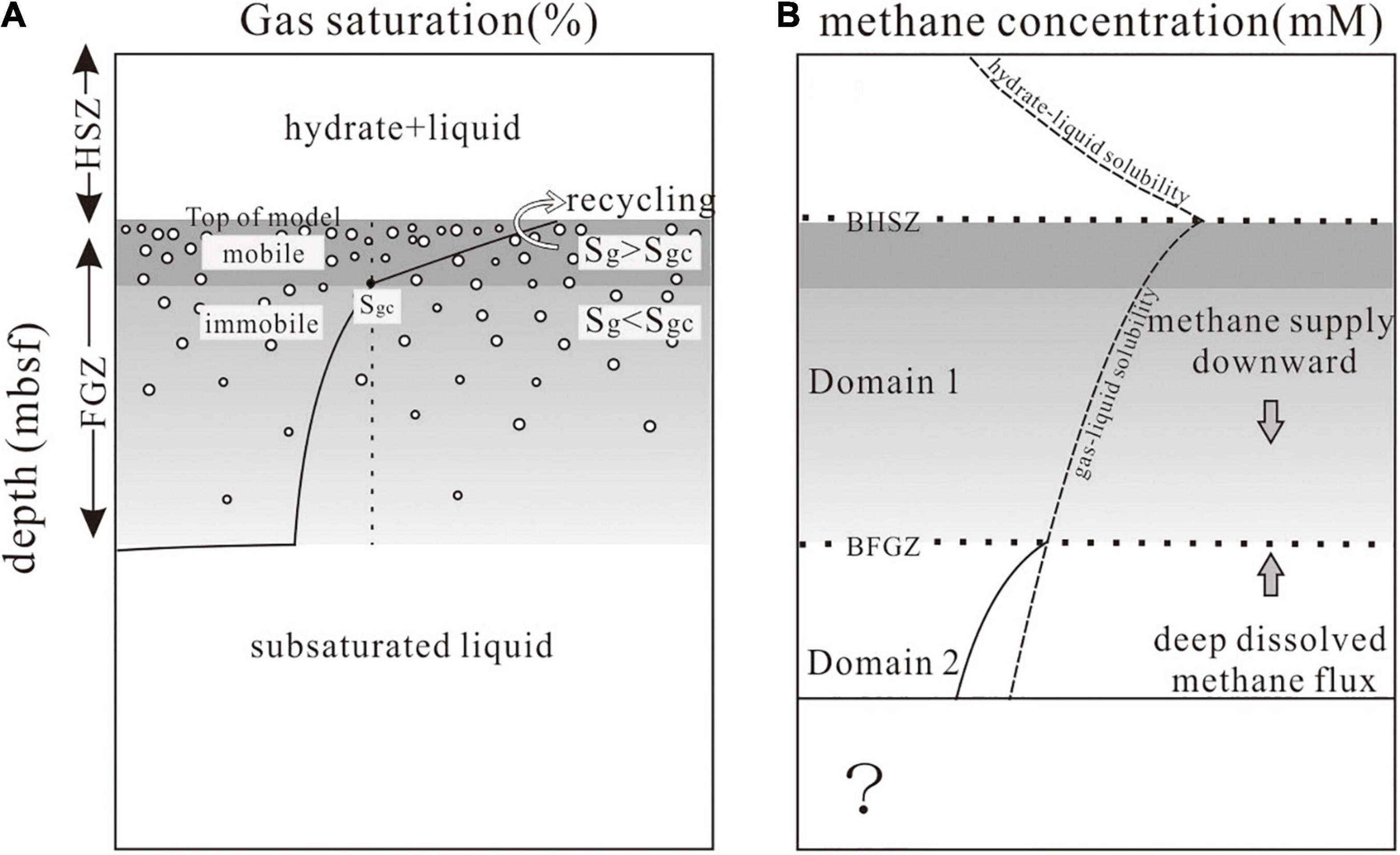
Frontiers A Numerical Model for Determining Deep Methane Flux

Explain how the compression factor varies with pressure and