The vapour pressure of a solution having 2.0 g of solute X (gram
4.9 (684) In stock
4.9 (684) In stock
The vapour pressure of a solution having 2.0 g of solute X (gram atomic mass=32 g/mol) in 100 g of CS2 (vapour pressure =854torr) is 848.9 torr.The molecular formula of solute 1) X 2)X2 3)X4 4)X8
The vapour pressure of a solution having 2-0 g of solute X -gram atomic mass-32 g-mol- in 100 g of CS2 -vapour pressure -854torr- is 848-9 torr-The molecular formula of solute 1- X 2-X2 3-X4 4-X8
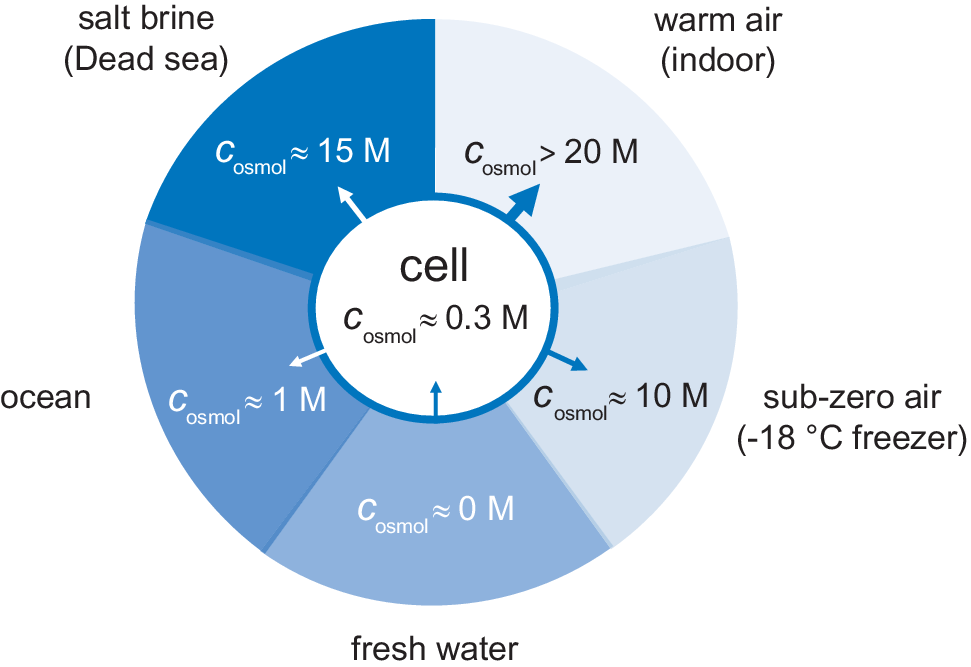
On the osmotic pressure of cells, QRB Discovery
Fluids, Free Full-Text

Solutions Homework Help, Questions with Solutions - Kunduz

The vapour pressure of a dilute aqueous solution of glucose is `700 mm` of `Hg` at `373 K`.
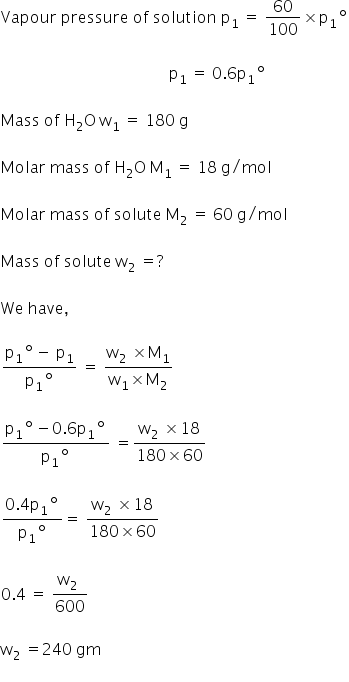
Calcualte the mass of a non volatile solute X that should be dissolved in 180g of water to reduce its vapor pressure to 60% where the molar mass of X is 60g/mol.

The vapour pressure of CS(2) at 50^(@)C is 854 torr and a solution o
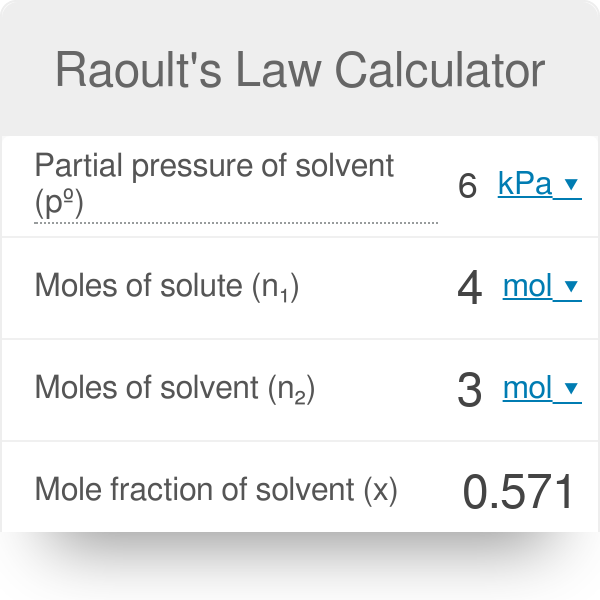
Raoult's Law Calculator

Comparison of the osmotic coefficient of the CaCl 2(aq) solution
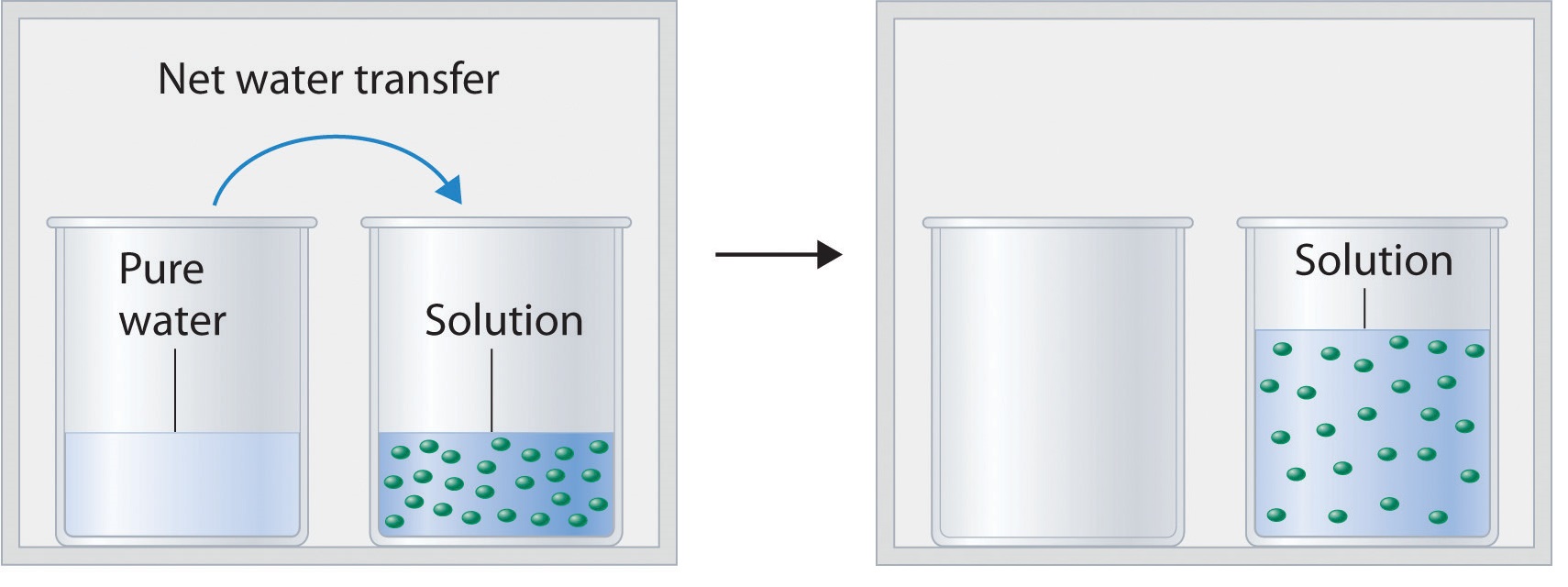
13.6: Vapor Pressures of Solutions - Chemistry LibreTexts
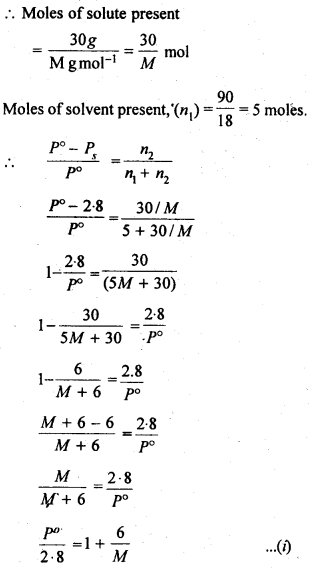
NCERT Solutions For Class 12 Chemistry Chapter 2 Solutions

Composition profiles x 1 z and x 2 z as functions of the distance from

Solutions Homework Help, Questions with Solutions - Kunduz

Calculate the mole fraction of toluene in the vapour phase which is in equilibrium with a solut

14. The vapour pressure of the solution having 2.0 g of solute (a molecu the solution having 2.0 g of solute (a molecule of x with atomic ma = 32 g/mol) in
When 2.5g of a non-volatile solute is added in 75g of water, the vapour pressure of the solution is 710mm of Hg. What will be the molar mass of the solute if